What is Qualitative Analysis ?

1. Qualitative analysis is a technique that is used to identify the cations and anions that are present in a substance.
2. The qualitative analysis methods that can be used on a salt are as follows:
a) analysis of the colour and physical properties of the salt
b) analysis of the solubility of the salt in water
c) analysis of the gases released
d) analysis of the action of heat on the salt
e) analysis of the confirmation tests for the anion
f) analysis of the confirmation tests for the cation
2. The qualitative analysis methods that can be used on a salt are as follows:
a) analysis of the colour and physical properties of the salt
b) analysis of the solubility of the salt in water
c) analysis of the gases released
d) analysis of the action of heat on the salt
e) analysis of the confirmation tests for the anion
f) analysis of the confirmation tests for the cation
Analysis of the colour and Physical Properties of a Salt

1. Analysis of the colour and physical properties is a preliminary step in the identification of a salt.
2. The physical properties of a salt include its physical state, whether it is a solid, liquid, gas or an aqueous solution.
3. The table below shows the colour of some salts.
2. The physical properties of a salt include its physical state, whether it is a solid, liquid, gas or an aqueous solution.
3. The table below shows the colour of some salts.
4.The colour of a solid or solution of a sa|t allows inferences to be made regarding the cations and anions that are present in the salt.
Analysis of the solubility of salts in water

1. Analysis of the solubility of a salt in water is easy to carry out in the laboratory.
2. Different salts have different levels of solubility in water and the presence of cations and anions in the salt can be predicted.
3. Analysis of the solubility of a salt in water can be carried out by dissolving a constant mass of the salt in a fixed volume of water.
4. The table below shows the solubility of some salts in water.
2. Different salts have different levels of solubility in water and the presence of cations and anions in the salt can be predicted.
3. Analysis of the solubility of a salt in water can be carried out by dissolving a constant mass of the salt in a fixed volume of water.
4. The table below shows the solubility of some salts in water.
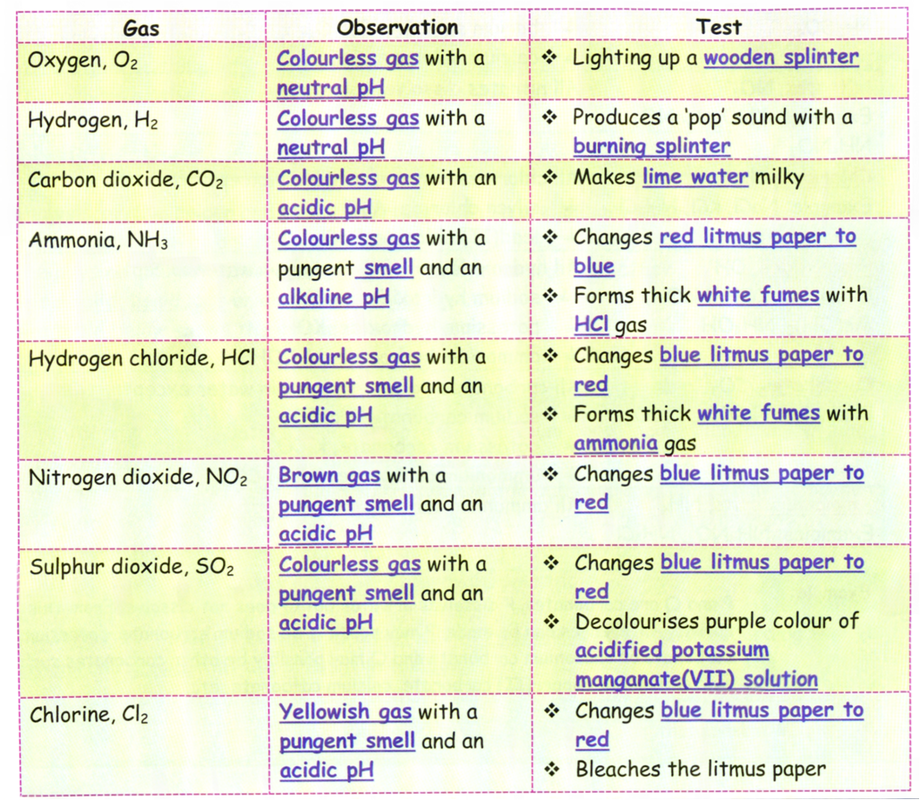
Analysis of Gases that are Released

1. Most salts will releases specific gases when:
a) they are heated
b) they reacts with an acid or alkali
2. The gases that are released can be identified by:
a) observing the colour
b) testing with blue litmus paper and red litmus paper
c) testing with lime water
d) testing with a wooden splinter
e) testing with a solution of acidified potassium manganate(Vll)
3. The table below shows the confirmatory tests for some gases that are released when some salts are heated.
a) they are heated
b) they reacts with an acid or alkali
2. The gases that are released can be identified by:
a) observing the colour
b) testing with blue litmus paper and red litmus paper
c) testing with lime water
d) testing with a wooden splinter
e) testing with a solution of acidified potassium manganate(Vll)
3. The table below shows the confirmatory tests for some gases that are released when some salts are heated.
Action of Heat on Salts
1. The action of heat on a salt is the first test which is normally carried out in a series of chemical tests in qualitative analysis.
2. When a salt is heated, a change in the colour of the salt occurs and the gas that is released can be observed.
2. When a salt is heated, a change in the colour of the salt occurs and the gas that is released can be observed.
3. The observations made from the heating process can be used to predict the cations and anions present in a salt.
4. All carbonates decompose when heated to release carbon dioxide except for potassium carbonate and sodium carbonate.
4. All carbonates decompose when heated to release carbon dioxide except for potassium carbonate and sodium carbonate.
5. All nitrates decompose when heated and release oxygen and nitrogen dioxide except for potassium nitrate and sodium nitrate which only release oxygen gas.
6. Most sulphates do not decompose when heated but some sulphates decompose when heated strongly to produce sulphur trioxide.
7. All chlorides do not decompose when heated except ammonium chloride.
Test for Anions in Aqueous Solution

1. The presence of anions in aqueous solution can be identified by carrying out :
a) reaction with acids
b) reaction with other chemicals
2. The table below shows the observations made on four different anions.
a) reaction with acids
b) reaction with other chemicals
2. The table below shows the observations made on four different anions.
Test for Cations in Aqueous Solution

1. The presence of cations in aqueous solution can be identified by carrying out:
a) reaction with sodium hydroxide solution
b) reaction with ammonia
2. In the form of aqueous solutions, the cations can be classified into cations are colourless in aqueous solution and cations which are coloured in solution.
a) Colourless = Cu2+, Ca2+, Zn2+, Al3+, Pb2+
b) Coloured = Cu2+, Fe2+, Fe3+
3. The table below shows the observations made in the presence of some cations in aqueous solution.
a) reaction with sodium hydroxide solution
b) reaction with ammonia
2. In the form of aqueous solutions, the cations can be classified into cations are colourless in aqueous solution and cations which are coloured in solution.
a) Colourless = Cu2+, Ca2+, Zn2+, Al3+, Pb2+
b) Coloured = Cu2+, Fe2+, Fe3+
3. The table below shows the observations made in the presence of some cations in aqueous solution.